Warning: include(common/header.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 22
Warning: include(): Failed opening 'common/header.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 22
Warning: include(common-home/leftcolumn_epa.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 30
Warning: include(): Failed opening 'common-home/leftcolumn_epa.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 30
EPA Methods List with Links
US EPA Method 107a Determination Of Vinyl Chloride Content Of Solvents, Resin-Solvent Solution, Polyvinyl Chloride Resin, Resin Slurry, Wet Resin, And Latex Samples
NOTE: This method does not include all of the specifications (e.g., equipment and supplies) and procedures (e.g., sampling and analytical) essential to its performance. Some material is incorporated by reference from other methods in this part. Therefore, to obtain reliable results, persons using this method should have a thorough knowledge of at least the following additional test methods: Method 106 and Method 107.
1.0 Scope and Application.
1.1 Analytes.
| Analyte | CAS No. | Sensitivity |
| Vinyl Chloride (CH2:CHCl) | 75-01-4 | Dependent upon analytical equipment |
1.2 Applicability.
This is an alternative method is applicable for the determination of the vinyl chloride content of solvents, resin solvent solutions, polyvinyl chloride (PVC) resin, wet cake slurries, latex, and fabricated resin samples. This method is not acceptable where methods from Section 304(h) of the Clean Water Act, 33 U.S.C. 1251 et seq., (the Federal Water Pollution Control Act Amendments of 1972 as amended by the Clean Water Act of 1977) are required.
1.3 Data Quality Objectives.
Adherence to the requirements of this method will enhance the quality of the data obtained from air pollutant sampling methods.
2.0 Summary of Method.
2.1 A sample of PVC is collected, and an aliquot is dissolved in either tetrahydrofuran or N,N- dimethylacetamide. The dissolved sample is injected directly into a chromatograph. All volatile material in the sample evaporates into the carrier gas stream of the chromatograph, and is analyzed for vinyl chloride content.
3.0 Definitions. [Reserved].
4.0 Interferences.
Same as Method 107, Section 4.0.
5.0 Safety.
5.1 Disclaimer.
This method may involve hazardous materials, operations, and equipment. This test method may not address all of the safety problems associated with its use. It is the responsibility of the user of this test method to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to performing this test method.
5.2 Toxic Analyte.
Same as Method 107, Section 5.2.
6.0 Equipment and Supplies.
6.1 Sample Collection.
The following equipment is required:
6.1.1 glass Bottles. 16-oz wide mouth wide polyethylene-lined, screw-on tops.
6.1.2 Adhesive Tape. To prevent loosening of bottle tops.
6.2 Sample Recovery.
The following equipment is required:
6.2.1 glass Vials. 20-ml capacity with polycone screw caps.
6.2.2 Analytical Balance. Capable of weighing to 0.01 gram.
6.2.3 Syringe. 50-microliter size, with removable needle.
6.2.4 Fritted glass Sparger. Fine porosity.
6.2.5 Aluminum Weighing Dishes.
6.2.6 Sample Roller or Shaker. To help dissolve sample.
6.3 Analysis.
The following equipment is required:
6.3.1 Gas Chromatograph. Hewlett Packard Model 5720A or equivalent.
6.3.2 Chromatograph Column. Stainless steel, 6.1 m by 3.2 mm, packed with 20 percent Tergitol E-35 on Chromosorb W AW 60/80 mesh. The analyst may use other columns provided that the precision and accuracy of the analysis of vinyl chloride standards are not impaired and that he has available for review information confirming that there is adequate resolution of the vinyl chloride peak. (Adequate resolution is defined as an area overlap of not more than 10 percent of the vinyl chloride peak by an interfering peak. Calculation of area overlap is explained in Procedure 1 of appendix C to this part: "Determination of Adequate Chromatographic Peak Resolution.")
6.3.3 Valco Instrument Six-Port Rotary Valve. For column back flush.
6.3.4 Septa. For chromatograph injection port.
6.3.5 Injection Port Liners. For chromatograph used.
6.3.6 Regulators. For required gas cylinders.
6.3.7 Soap Film flowmeter. Hewlett Packard No. 0101-0113 or equivalent.
6.4 calibration.
The following equipment is required:
6.4.1 Analytical Balance. Capable of weighing to 0.0001 g.
6.4.2 Erlenmeyer Flask With glass Stopper. 125 ml.
6.4.3 Pipets. 0.1, 0.5, 1, 5, 10, and 50 ml.
6.4.4 Volumetric Flasks. 10 and 100 ml.
7.0 Reagents and Standards.
7.1 Analysis.
Same as Method 106, Section 7.1, with the addition of either of the following:
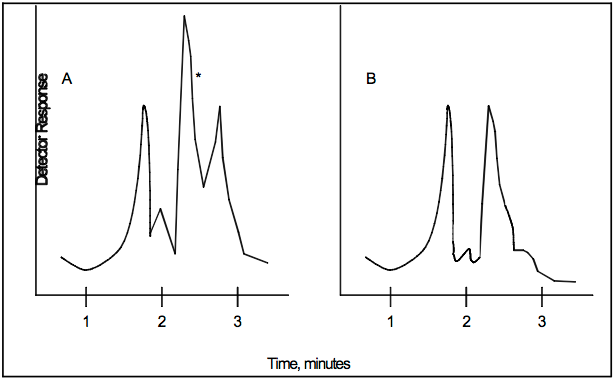
7.1.1 Tetrahydrofuran (THF). Reagent grade. Analyze the THF by injecting 10 microliters into the prepared gas chromatograph. Compare the THF chromatogram with that shown in Figure 107A-1. If the chromatogram is comparable to A, the THF should be sparged with pure nitrogen for approximately 2 hours using the fritted glass sparger to attempt to remove the interfering peak. Reanalyze the sparged THF to determine whether the THF is acceptable for use. If the scan is comparable to B, the THF should be acceptable for use in the analysis.
7.1.2 N, N-Dimethylacetamide (DMAC). Spectrographic grade. For use in place of THF.
7.2 calibration.
Vinyl chloride, same as described in Method 106, Section 7.2.1, is required.
8.0 Sample Collection, Preservation, Storage, and Transport.
8.1 Sample Collection.
Allow the liquid or dried resin to flow from a tap on the tank, silo, or pipeline until the tap has been purged. Fill a wide-mouth pint bottle, and immediately tightly cap the bottle. Place an identifying label on each bottle and record the date, time, sample location, and material.
8.2 Sample Storage.
All samples must be analyzed within 24 hours of collection, and must be refrigerated during this period.
9.0 Quality Control.
| Section | Quality Control Measure | Effect |
| 10.2 | Chromatograph calibration | Ensure precision and accuracy of chromatograph |
| 11.3.1 | Repetitive sample analyses | Ensure precision of analyses |
10.0 Calibration and Standardization.
10.1 Preparation of Standards.
Prepare a 1 percent by weight (approximate) solution of vinyl chloride in THF or DMAC by bubbling vinyl chloride gas from a cylinder into a tared 125-ml glass-stoppered flask containing THF or DMAC. The weight of vinyl chloride to be added should be calculated prior to this operation, i.e., 1 percent of the weight of THF or DMAC contained in the tared flask. This must be carried out in a laboratory hood. Adjust the vinyl chloride flow from the cylinder so that the vinyl chloride dissolves essentially completely in the THF or DMAC and is not blown to the atmosphere. Take particular care not to volatize any of the solution. Stopper the flask and swirl the solution to effect complete mixing. Weigh the stoppered flask to nearest 0.1 mg to determine the exact amount of vinyl chloride added. Pipet 10 ml of the approximately 1 percent solution into a 100-ml glass-stoppered volumetric flask, and add THF or DMAC to fill to the mark. Cap the flask and invert 10 to 20 times. This solution contains approximately 1,000 ppm by weight of vinyl chloride (note the exact concentration). Pipet 50-, 10-, 5-, 1-, 0.5-, and 0.1-ml aliquots of the approximately 1,000 ppm solution into 10 ml glass stoppered volumetric flasks. Dilute to the mark with THF or DMAC, cap the flasks and invert each 10 to 20 times. These solutions contain approximately 500, 100, 50, 10, 5, and 1 ppm vinyl chloride. Note the exact concentration of each one. These standards are to be kept under refrigeration in stoppered bottles, and must be renewed every 3 months.
10.2 Preparation of Chromatograph calibration Curve.
Obtain the GC readout for each of the six final solutions prepared in Section 10.1 by using the procedure in Section 11.3.1. Prepare a chart plotting peak height obtained from the chromatogram of each solution versus the known concentration. Draw a straight line through the points derived by the least squares method.
11.0 Analytical Procedure.
11.1 Preparation of GC.
Install the chromatographic column, and condition overnight at 70 °C (160 °F). Do not connect the exit end of the column to the detector while conditioning.
11.1.1 flow Rate Adjustments.
Adjust the flow rate as follows:
11.1.1.1 Nitrogen Carrier Gas. Set regulator on cylinder to read 60 psig. Set column flow controller on the chromatograph using the soap film flowmeter to yield a flow rate of 40 cc/min.
11.1.1.2 Burner Air Supply. Set regulator on the cylinder at 40 psig. Set regulator on the chromatograph to supply air to the burner to yield a flow rate of 250 to 300 cc/min using the flowmeter.
11.1.1.3 Hydrogen. Set regulator on cylinder to read 60 psig. Set regulator on the chromatograph to supply 30 to 40 cc/min using the flowmeter. Optimize hydrogen flow to yield the most sensitive detector response without extinguishing the flame. Check flow with flowmeter and record this flow.
11.1.1.4 Nitrogen Back Flush Gas. Set regulator on the chromatograph using the soap film flowmeter to yield a flow rate of 40 cc/min.
11.1.2 temperature Adjustments.
Set temperature as follows:
11.1.2.1 oven (chromatographic column) at 70 °C (160 °F).
11.1.2.2 Injection Port at 100 °C (212 °F).
11.1.2.3 Detector at 300 °C (570 °F).
11.1.3 Ignition of Flame Ionization Detector. Ignite the detector according to the manufacturer's instructions. Allow system to stabilize approximately 1 hour.
11.1.4 Recorder. Set pen at zero and start chart drive.
11.1.5 Attenuation. Set attenuation to yield desired peak height depending on sample VCM content.
11.2 Sample Treatment.
All samples must be recovered and analyzed within 24 hours after collection.
11.2.1 Resin Samples.
Weigh 9.00 0.01 g of THF or DMAC in a tared 20-ml vial. Add 1.00 0.01 g of resin to the tared vial containing the THF or DMAC. Close the vial tightly with the screw cap, and shake or otherwise agitate the vial until complete solution of the resin is obtained. Shaking may require several minutes to several hours, depending on the nature of the resin.
11.2.2 Suspension Resin Slurry and Wet Resin Sample.
Slurry must be filtered using a small Buchner funnel with vacuum to yield a wet resin sample. The filtering process must be continued only as long as a steady stream of water is exiting from the funnel. Excessive filtration time could result in some loss of VCM. The wet resin sample is weighed into a tared 20-ml vial with THF or DMAC as described earlier for resin samples (Section 11.2.1) and treated the same as the resin sample. A sample of the wet resin is used to determine total solids as required for calculating the residual VCM (Section 11.5).
11.2.3 Latex and Resin Solvent Solutions.
Samples must be thoroughly mixed. Weigh 1.00 0.01 g of the latex or resin-solvent solution into a 20-ml vial containing 9.00 0.01 g of THF or DMAC as for the resin samples (Section 11.2.1). Cap and shake until complete solution is obtained. Determine the total solids of the latex or resin solution sample (Section 11.5).
11.2.4 Solvents and Non-viscous Liquid Samples.
No preparation of these samples is required. The neat samples are injected directly into the GC.
11.3 Chromatographic Analyses.
11.3.1 Sample Injection.
Remove needle from 50-microliter syringe. Open sample vial and draw 50-microliters of THF or DMAC sample recovery solution into the syringe. Recap sample vial. Attach needle to the syringe and while holding the syringe vertically (needle uppermost), eject 40 microliters into an absorbent tissue. Wipe needle with tissue. Now inject 10 microliters into chromatograph system. Repeat the injection until two consecutive values for the height of the vinyl chloride peak agree within 5 percent of their average. Use the average value for these two peak heights to compute the sample concentration.
11.3.2 Back Flush. After 4 minutes has elapsed after sample injection, actuate the back flush valve to purge the first 4 feet of the chromatographic column of solvent and other high boilers.
11.3.3 Sample Data. Record on the chromatograph strip chart the data from the sample label.
11.3.4 Elution Time. Vinyl chloride elutes at 2.8 minutes. Acetaldehyde elutes at 3.7 minutes. Analysis is considered complete when chart pen becomes stable. After 5 minutes, reset back flush valve and inject next sample.
11.4 Chromatograph Servicing.
11.4.1 Septum. Replace after five sample injections.
11.4.2 Sample Port Liner. Replace the sample port liner with a clean spare after five sample injections.
11.4.3 Chromatograph Shutdown. If the chromatograph has been shut down overnight, rerun one or more samples from the preceding day to test stability and precision prior to starting on the current day's work.
11.5 Determination of Total Solids (TS).
11.5.1 For wet resin, resin solution, and PVC latex samples, determine the TS for each sample by accurately weighing approximately 3 to 5 grams of sample into a tared aluminum pan. The initial procedure is as follows:
11.5.1.1 Where water is the major volatile component:
Tare the weighing dish, and add 3 to 5 grams of sample to the dish. Weigh to the nearest milligram.
11.5.1.2 Where volatile solvent is the major volatile component: Transfer a portion of the sample to a 20-ml screw cap vial and cap immediately. Weigh the vial to the nearest milligram. Uncap the vial and transfer a 3- to 5-gram portion of the sample to a tared aluminum weighing dish. Recap the vial and reweigh to the nearest milligram. The vial weight loss is the sample weight.
11.5.2 To continue, place the weighing pan in a 130 °C oven for 1 hour. Remove the dish and allow to cool to room temperature in a desiccator. Weigh the pan to the nearest 0.1 mg. Total solids is the weight of material in the aluminum pan after heating divided by the net weight of sample added to the pan originally times 100.
12.0 Calculations and Data Analysis
12.1 Nomenclature.
| Cc | = | Concentration of vinyl chloride in the standard sample, ppm. |
| Crvc | = | Concentration of residual vinyl chloride monomer, |
| Hc | = | Peak height of the standard sample, mm. ppm. |
| Hs | = | Peak height of sample, mm. |
| Rf | = | Chromatograph response factor, ppm/mm. |
| TS | = | Total solids in the sample, weight fraction. |
| 0.888 | = | Specific gravity of THF. |
12.2 Response Factor.
From the calibration curve described in Section 10.2, select the value of Cc that corresponds to Hc for each sample. Compute the response factor, Rf, for each sample as follows:
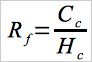 Eq. 107A-1
Eq. 107A-1
12.3 Residual vinyl chloride monomer concentration (Crvc) or vinyl chloride monomer concentration in resin:
 Eq. 107A-2
Eq. 107A-2
12.4 Samples containing volatile material, i.e., resin solutions, wet resin, and latexes:
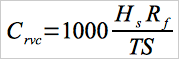 Eq. 107A-3
Eq. 107A-3
12.5 Samples of solvents and in process wastewater:
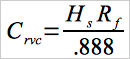 Eq. 107A-4
Eq. 107A-4
13.0 Method Performance.
13.1 Range and Sensitivity.
The lower limit of detection of vinyl chloride in dry PVC resin is 0.2 ppm. For resin solutions, latexes, and wet resin, this limit rises inversely as the nonvolatile (resin) content decreases. With proper calibration, the upper limit may be extended as needed.
14.0 Pollution Prevention. [Reserved]
15.0 Waste Management. [Reserved]
16.0 References.
Same as Method 107, Section 16, with the addition of the following:
1. Communication from R.N. Wheeler, Jr., Union Carbide Corporation. Part 61 National Emissions Standards for Hazardous Air Pollutants, Appendix B, Method 107 - Alternate Method. September 19, 1977.
17.0 Tables, Diagrams, flowcharts, and Validation Data.
Figure 107A-1. Example Chromatograms.
Warning: include(right-column/Services.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 234
Warning: include(): Failed opening 'right-column/Services.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 234
Warning: include(right-column/Products.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 235
Warning: include(): Failed opening 'right-column/Products.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 235
Warning: include(right-column/Resources.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 236
Warning: include(): Failed opening 'right-column/Resources.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 236
Warning: include(right-column/Experts.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 237
Warning: include(): Failed opening 'right-column/Experts.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 237
Warning: include(common-epa/right_col_method_ads.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 240
Warning: include(): Failed opening 'common-epa/right_col_method_ads.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 240
Warning: include(right-column/UniqueValue.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 241
Warning: include(): Failed opening 'right-column/UniqueValue.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 241
Warning: include(right-column/QualityCheck.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 242
Warning: include(): Failed opening 'right-column/QualityCheck.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 242
Warning: include(common/allstats.php): Failed to open stream: No such file or directory in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 249
Warning: include(): Failed opening 'common/allstats.php' for inclusion (include_path='.:/opt/homebrew/Cellar/php/8.3.4/share/php/pear') in /Users/lowerlevel/Sites/D7036/epamethods/air-test-methods/m-107a.php on line 249